About
About ENDOMETRIN
Prescribe ENDOMETRIN for your patients to support embryo implantation and early pregnancy by supplementation of corpus luteal function as part of an assisted reproductive technology (ART) treatment program for infertile women.1

In a US IVF trial with more than 1,200 patients, ENDOMETRIN was proven safe and effective.2
Study Design
ENDOMETRIN has been a part of IVF protocols since 2007.1
Support
Administration
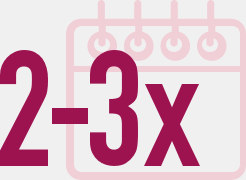
Adaptable dosing
Each ENDOMETRIN insert supplies 100 mg of progesterone and comes with its own single-use disposable applicator for sterile insertion into the vagina, 2 or 3 times a day, for up to 10 weeks, as specified.1

How to Use ENDOMETRIN
For full step-by-step instructions on how to use ENDOMETRIN, download the how-to guide for your patients.1


